1 Global CGT CDMO Market Outlook
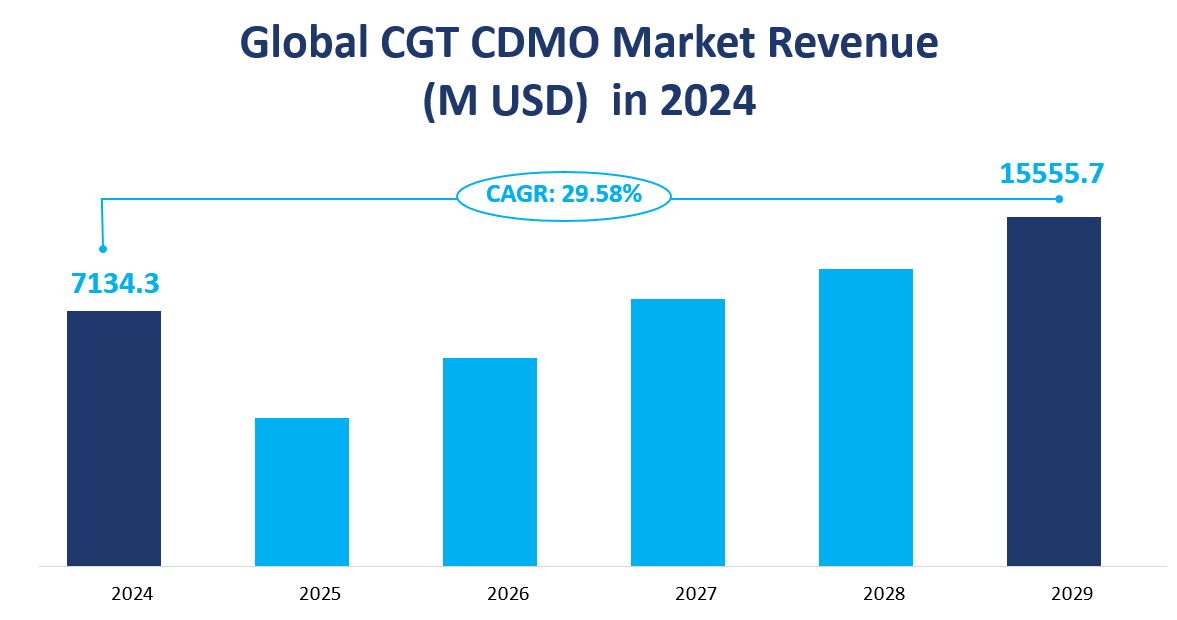
In 2024, the CGT CDMO market was anticipated to reached a substantial value, with an estimated total of $7,134.3 million. This figure reflected a CAGR of 29.58% from 2024 to 2029, indicating a period of rapid expansion for the industry. The CGT CDMO sector is a specialized segment within the pharmaceutical industry, focusing on the development and manufacturing of cell and gene therapies. These therapies involve the use of living cells, tissues, viral vectors, and non-viral gene modification components, which present unique challenges in research and production due to their complexity and sensitivity.
Figure Global CGT CDMO Market Revenue and CAGR 2024-2029

2 Drivers and Limitations of the CGT CDMO Market
The growth of the CGT CDMO market is propelled by several key factors. The development of the cell and gene therapy industry is a primary driver, with a growing burden of cancer and other targeted diseases leading to an increased demand for personalized and effective treatments. The rapid expansion of research on CGTs, substantial funding by venture capitalists, and technological innovations are also contributing to the market’s growth.
Additionally, the robust CGT pipeline, with an increasing number of product approvals and strong support from large pharmaceutical and biotechnology companies, is set to boost the demand for CGT CDMO services.
However, the market faces limitations that could hinder its growth. The manufacturing of CGTs is labor-intensive and expensive, with significant variation in product yield. Many patients may not be suitable candidates for CGTs due to pre-exposure to viral vectors, poor gene expression, or the development of immunogenicity. Furthermore, the production of gene therapies requires materials of biological origin, which can contain trace levels of process-related impurities that may trigger immunogenicity in patients. The complexity of analytical characterization of advanced therapy medicinal products (ATMPs) and the need for full traceability of materials from animal or human sources add to the challenges.
These factors, along with the potential for adverse events and the high costs associated with meeting stringent regulatory requirements, pose significant limitations to the market’s growth.
3 Technological Innovations in the CGT CDMO Market
The CGT CDMO market is characterized by continuous technological innovation and strategic mergers and acquisitions (M&A) that shape its landscape. Technological advancements in gene editing, such as CRISPR/Cas9, have opened new avenues for the development of gene therapies, requiring CDMOs to adapt and invest in cutting-edge manufacturing capabilities. The industry is also witnessing a trend where CDMOs are expanding their service offerings to provide a full suite of solutions, from drug discovery to commercialization, to meet the growing demand for one-stop-shop services.
4 Global CGT CDMO Market Analysis by Type
The CGT CDMO market encompasses several product types, each with its unique characteristics and applications. The main product types include Immune Cells, Stem Cells, Viral Vectors, and Plasmid DNA.
Immune Cells: These therapies involve the use of immune cells, such as T-cells, NK cells, and dendritic cells, which are engineered to target and destroy diseased cells. This includes well-known approaches like CAR-T cell therapies.
Stem Cells: Stem cell therapies utilize the regenerative potential of stem cells, which can differentiate into various cell types, to treat a range of diseases. This category includes both adult and pluripotent stem cells.
Viral Vectors: Viral vector therapies use modified viruses to deliver genetic material into cells, enabling the correction or modulation of genetic disorders. Examples include AAV, lentivirus, and adenovirus vectors.
Plasmid DNA: Plasmid DNA is used as a vector for gene therapies and DNA vaccines, allowing for the introduction of therapeutic genes into the body.
Immune Cells segment reached a value of $2,498.9 million in 2024. This dominance can be attributed to the significant advancements in immunotherapy, particularly in CAR-T cell technology, which has shown remarkable success in treating certain types of cancer.
Viral Vectors had the fastest growth rate, with a market value of $2,627.7 million in 2024. The rapid growth of this segment was driven by the increasing application of viral vectors in gene therapy, which offered a versatile and effective method for delivering therapeutic genes to target cells.
The growth of the Plasmid DNA segment was also noteworthy, with a market value of $1,022.2 million in 2024. The interest in plasmid DNA as a vector for gene therapies and vaccines has been growing, especially with the development of new technologies that facilitate the design and production of customized plasmids for various therapeutic targets.
Table Global CGT CDMO Market Value and Share by Type in 2024
| Product Type | Market Value (M USD) 2024 | Market Share 2024 (%) |
|---|---|---|
| Immune Cells | 2,498.9 | 35.03 |
| Stem Cells | 985.5 | 13.81 |
| Viral Vectors | 2,627.7 | 36.86 |
| Plasmid DNA | 1,022.2 | 14.34 |
5 Global CGT CDMO Market Analysis by Application
The CGT CDMO market serves various applications, each with distinct needs and implications for the field of healthcare. The primary applications include Pharmaceutical and Biotechnology Companies, Research and Academic Institutions.
Pharmaceutical and Biotechnology Companies encompasses companies that are focused on the research, development, and commercialization of cell and gene therapies. These entities require CGT CDMO services for process development, manufacturing, and compliance with regulatory standards.
Research and Academic Institutions includes universities, research institutes, and academic centers engaged in the exploration of cell and gene therapies. They often collaborate with CGT CDMOs for specialized manufacturing and development services that are not readily available in academic settings.
Pharmaceutical and Biotechnology Companies hold the largest market share, with a market value of $6,396.5 million in 2024. This dominance was attributed to the significant investment in R&D by these companies and the commercial potential of the therapies they develop.
The fastest-growing application was Research and Academic Institutions, with a market value of $627.7 million in 2024. The rapid growth in this sector can be attributed to the increasing number of academic institutions engaging in cutting-edge research in cell and gene therapies, often in collaboration with industry partners.
Table Global CGT CDMO Market Value and Share by Application in 2024
| Application | Market Value (M USD) 2024 | Market Share 2024 (%) |
|---|---|---|
| Pharmaceutical and Biotechnology Companies | 6,396.5 | 89.71 |
| Research and Academic Institutions | 627.7 | 8.77 |
6 Global CGT CDMO Market Analysis by Region
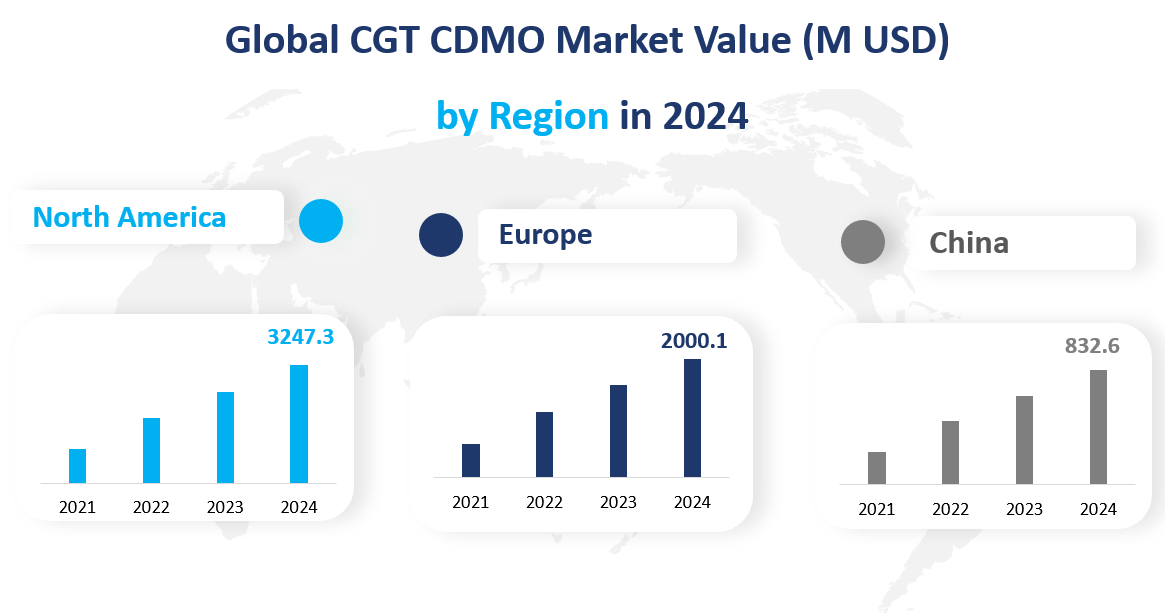
In 2024, the global CGT CDMO market reflected a dynamic landscape with distinct regional performances.
North America: North America concluded 2024 as the largest regional market for CGT CDMO services, with a market value of $3247.3 million. The region’s dominance was attributed to its robust pharmaceutical industry, advanced healthcare infrastructure, and a supportive regulatory environment. The United States, in particular, was home to many leading CGT companies and a hub for biotechnology innovation.
Europe: Europe’s market value reached $2000.1 million in 2024. The region’s mature pharmaceutical market, along with a strong focus on research and development, contributed to its significant share in the global CGT CDMO market. Additionally, the region’s collaborative approach to drug development and regulatory harmonization supported market growth.
China: China’s market value grew to $832.6 million in 2024, positioning it as a key player in the global CGT CDMO landscape. The country’s rapid economic growth, increasing investment in biotechnology, and supportive government policies created a conducive environment for the expansion of the CGT sector.
China was identified as the fastest-growing region in the CGT CDMO market in 2024, with a growth rate of 40.02%. The rapid growth in China’s market was attributed to the country’s strategic investments in biotechnology, its large patient population offering diverse clinical trial opportunities, and government initiatives to bolster the domestic pharmaceutical industry.
Figure Global CGT CDMO Market Value (M USD) by Region in 2024

7 Global CGT CDMO Market Analysis by Top 5 Players
7.1 Catalent
Company Introduction and Business Overview:
Catalent is a leading global provider of advanced delivery technologies and development solutions for drugs, biologics, gene therapies, and consumer health products. Established in 2007 and headquartered in Somerset, New Jersey, Catalent operates worldwide, offering a comprehensive portfolio of services from drug discovery to commercialization.
Products Offered:
Catalent’s product offerings include a wide range of cell therapy services, such as CAR-T, TCR, TILs, NK cells, iPSCs, and MSCs. They also specialize in viral vector development and manufacturing for gene therapies, including plasmid DNA, adeno-associated viral (AAV), lentiviral, and other viral vectors, as well as oncolytic viruses and live virus vaccines.
7.2 Lonza
Company Introduction and Business Overview:
Lonza, established in 1897 and headquartered in Basel, Switzerland, is a Swiss multinational manufacturing company serving the pharmaceutical, biotechnology, and nutrition sectors. Lonza provides product development services and custom manufacturing of biopharmaceuticals, detection systems, and services for the bioscience sector.
Products Offered:
Lonza’s product portfolio focuses on cell and gene therapies, offering integrated CDMO services across the full value chain of these modalities, including allogeneic and autologous therapies and viral vectors.
7.3 Thermo Fisher Scientific
Company Introduction and Business Overview:
Thermo Fisher Scientific, founded in 1956 and based in Waltham, Massachusetts, is a leading American supplier of scientific instrumentation, reagents and consumables, and software services. The company has a global presence and has acquired several reagent, consumable, instrumentation, and service providers, expanding its capabilities in the CGT CDMO space.
Products Offered:
Thermo Fisher provides integrated solutions to support cell therapy development at every stage, from discovery through clinical trials to commercialization. Their offerings include cell therapy ancillary materials, cell-based immunotherapy solutions, stem cell therapy solutions, and manufacturing support.
7.4 Charles River Laboratories
Company Introduction and Business Overview:
Charles River Laboratories, established in 1947 and headquartered in Wilmington, Massachusetts, is a global pharmaceutical company specializing in preclinical and clinical laboratory services, gene therapy, and cell therapy services for the pharmaceutical, medical device, and biotechnology industries.
Products Offered:
Charles River’s product and service offerings include cell therapy manufacturing services, viral vector manufacturing, plasmid DNA manufacturing services, and a range of other services to support the development and commercialization of cell and gene therapies.
7.5 WuXi AppTec
Company Introduction and Business Overview:
WuXi AppTec, founded in 2000 and headquartered in Shanghai, China, operates globally and provides a broad portfolio of R&D and manufacturing services across Asia, Europe, and North America. The company offers integrated, end-to-end services, including chemistry drug CRDMO, biology discovery, preclinical testing, clinical research services, and cell and gene therapies CTDMO.
Products Offered:
WuXi AppTec’s CTDMO services are tailored to meet specific needs, covering viral vectors for gene therapies, autologous and allogeneic cell therapies, gene-mediated cell therapies like CAR-T, and emerging technologies such as CRISPR/Cas9, oncolytic viruses, NK cells, and exosomes.
1 CGT CDMO Introduction and Market Overview
1.1 Objectives of the Study
1.2 Overview of CGT CDMO
1.3 CGT CDMO Market Scope and Market Size Estimation
1.3.1 Market Concentration Ratio and Market Maturity Analysis
1.3.2 Global CGT CDMO Value and Growth Rate from 2019-2029
1.4 Market Segmentation
1.4.1 Types of CGT CDMO
1.4.2 Applications of CGT CDMO
1.4.3 Research Regions
1.5 Market Dynamics
1.5.1 Drivers
1.5.2 Limitations
1.5.3 Opportunities
1.5.4 Trends
1.6 Industry News and Policies by Regions
1.6.1 Industry News
1.6.2 Industry Policies
1.7 CGT CDMO Industry Development Trends under COVID-19 Outbreak
1.7.1 Global COVID-19 Status Overview
1.7.2 Influence of COVID-19 Outbreak on CGT CDMO Industry Development
1.8 Global CGT CDMO Value and Market Share by Research Phase (2019-2029)
1.9 Global CGT CDMO Value and Market Share by Service Type by Region (2019-2029)
1.10 Impact of Russia and Ukraine War
2 Industry Chain Analysis
2.1 Major Players of CGT CDMO
2.1.1 Major Players Business Distribution Regions of CGT CDMO in 2024
2.2 CGT CDMO Business Cost Structure Analysis
2.2.1 CGT CDMO Costs and Control Analysis
2.2.2 Labor Cost of CGT CDMO
2.3 Market Value Chain Analysis of CGT CDMO
2.4 Major Downstream Customers of CGT CDMO Analysis by Service Type by Region
3 Global CGT CDMO Market, by Type
3.1 Global CGT CDMO Value and Market Share by Type (2019-2024)
3.2 Global CGT CDMO Value and Growth Rate by Type (2019-2024)
3.2.1 Global CGT CDMO Value and Growth Rate of Immune Cells
3.2.2 Global CGT CDMO Value and Growth Rate of Stem Cells
3.2.3 Global CGT CDMO Value and Growth Rate of Viral Vectors
3.2.4 Global CGT CDMO Value and Growth Rate of Plasmid DNA
4 CGT CDMO Market, by Application
4.1 Downstream Market Overview
4.2 Global CGT CDMO Consumption Value and Market Share by Application (2019-2024)
4.3 Global CGT CDMO Consumption and Growth Rate by Application (2019-2024)
4.3.1 Global CGT CDMO Value and Growth Rate of Pharmaceutical and Biotechnology Companies (2019-2024)
4.3.2 Global CGT CDMO Value and Growth Rate of Research and Academic Institutions (2019-2024)
5 Global CGT CDMO Value by Region (2019-2024)
5.1 Global CGT CDMO Value and Market Share by Region (2019-2024)
5.2 Global CGT CDMO Value and Gross Margin (2019-2024)
5.3 North America CGT CDMO Value and Gross Margin (2019-2024)
5.3.1 North America CGT CDMO Market Under COVID-19
5.4 Europe CGT CDMO Value and Gross Margin (2019-2024)
5.4.1 Europe CGT CDMO Market Under COVID-19
5.5 China CGT CDMO Value and Gross Margin (2019-2024)
5.5.1 China CGT CDMO Market Under COVID-19
5.6 Japan CGT CDMO Value and Gross Margin (2019-2024)
5.6.1 Japan CGT CDMO Market Under COVID-19
5.7 Middle East and Africa CGT CDMO Value and Gross Margin (2019-2024)
5.7.1 Middle East and Africa CGT CDMO Market Under COVID-19
5.8 India CGT CDMO Value and Gross Margin (2019-2024)
5.8.1 India CGT CDMO Market Under COVID-19
5.9 South America CGT CDMO Value and Gross Margin (2019-2024)
5.9.1 South America CGT CDMO Market Under COVID-19
6 Competitive Landscape
6.1 Competitive Profile
6.1.1 Major Players Value Share (2019-2024)
6.2 Catalent
6.2.1 Company Profiles
6.2.2 CGT CDMO Service Introduction
6.2.3 Catalent Value, Gross and Gross Margin 2019-2024
6.3 Lonza
6.3.1 Company Profiles
6.3.2 CGT CDMO Service Introduction
6.3.3 Lonza Value, Gross and Gross Margin 2019-2024
6.4 Thermo Fisher
6.4.1 Company Profiles
6.4.2 CGT CDMO Service Introduction
6.4.3 Thermo Fisher Value, Gross and Gross Margin 2019-2024
6.5 Charles River
6.5.1 Company Profiles
6.5.2 CGT CDMO Service Introduction
6.5.3 Charles River Value, Gross and Gross Margin 2019-2024
6.6 WuXi AppTec
6.6.1 Company Profiles
6.6.2 CGT CDMO Service Introduction
6.6.3 WuXi AppTec Value, Gross and Gross Margin 2019-2024
6.7 Oxford Biomedica (OXB)
6.7.1 Company Profiles
6.7.2 CGT CDMO Service Introduction
6.7.3 Oxford Biomedica (OXB) Value, Gross and Gross Margin 2019-2024
6.8 GenScript
6.8.1 Company Profiles
6.8.2 CGT CDMO Service Introduction
6.8.3 GenScript Value, Gross and Gross Margin 2019-2024
6.9 Novartis
6.9.1 Company Profiles
6.9.2 CGT CDMO Service Introduction
6.9.3 Novartis Value, Gross and Gross Margin 2019-2024
6.10 Pharmaron
6.10.1 Company Profiles
6.10.2 CGT CDMO Service Introduction
6.10.3 Pharmaron Value, Gross and Gross Margin 2019-2024
6.11 Porton
6.11.1 Company Profiles
6.11.2 CGT CDMO Service Introduction
6.11.3 Porton Value, Gross and Gross Margin 2019-2024
7 Global CGT CDMO Market Analysis and Forecast by Type and Application
7.1 Global CGT CDMO Market Value Forecast, by Type (2024-2029)
7.1.1 Immune Cells Market Value Forecast (2024-2029)
7.1.2 Stem Cells Market Value Forecast (2024-2029)
7.1.3 Viral Vectors Market Value Forecast (2024-2029)
7.1.4 Plasmid DNA Market Value Forecast (2024-2029)
7.2 Global CGT CDMO Market Value Forecast, by Application (2024-2029)
7.2.1 Pharmaceutical and Biotechnology Companies Market Value Forecast (2024-2029)
7.2.2 Research and Academic Institutions Market Value Forecast (2024-2029)
8 CGT CDMO Market Analysis and Forecast by Region
8.1 North America Market Value Forecast (2024-2029)
8.2 Europe Market Value Forecast (2024-2029)
8.3 China Market Value Forecast (2024-2029)
8.4 Japan Market Value Forecast (2024-2029)
8.5 Middle East and Africa Market Value Forecast (2024-2029)
8.6 India Market Value Forecast (2024-2029)
8.7 South America Market Value Forecast (2024-2029)
8.8 CGT CDMO Market Forecast Under COVID-19
9 New Project Feasibility Analysis
9.1 Industry Barriers and New Entrants SWOT Analysis
10 Research Finding and Conclusion
11 Appendix
11.1 Methodology
11.2 Research Data Source
