1 Global Standard to Auto-Disable Syringe Market Scope
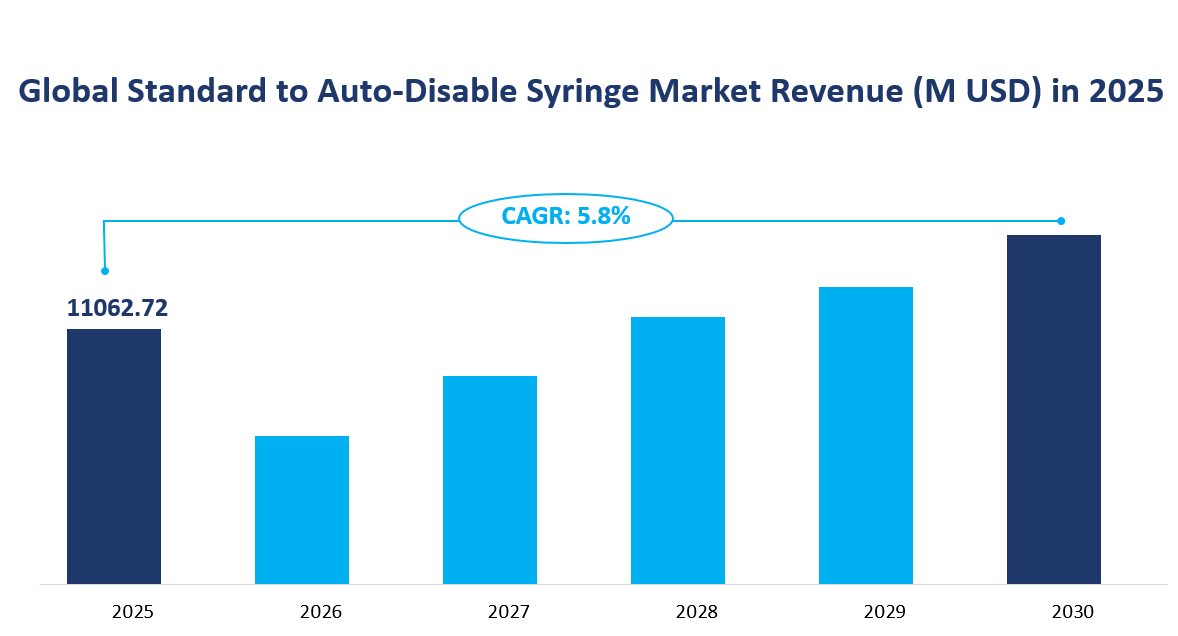
The global Standard to Auto-Disable Syringe market is projected to reach a total value of 11,062.72 million USD by 2025. The Compound Annual Growth Rate (CAGR) of the market is estimated to be 5.8% from 2025 to 2030. Standard to Auto-Disable Syringes are medical devices designed to prevent the reuse of syringes, thereby reducing the risk of infection and ensuring patient safety. These syringes are widely used in various healthcare settings, including public hospitals, private clinics, and nursing homes.
Figure Global Standard to Auto-Disable Syringe Market Revenue and CAGR 2025-2030

2 Drivers and Constraints of Standard to Auto-Disable Syringe Market
The growth of the Standard to Auto-Disable Syringe market is driven by several key factors. Firstly, the increasing global population and the rise in healthcare demand are driving the need for safe and reliable medical devices. The use of Standard to Auto-Disable Syringes helps prevent the spread of infectious diseases, making them an essential component in healthcare facilities. Secondly, the growing awareness of healthcare professionals and patients about the risks associated with the reuse of syringes is leading to a higher adoption rate of these devices. Additionally, technological advancements in the manufacturing of syringes have improved their quality and reliability, further boosting market growth.
However, the market also faces several constraints. One of the main challenges is the high cost of production, which can limit the affordability and accessibility of these syringes in some regions. The need for strict regulatory compliance also adds to the production costs and complexity. Furthermore, the market is highly competitive, with several established players and new entrants vying for market share. This competition can lead to price pressures and reduced profit margins for manufacturers.
3 Standard to Auto-Disable Syringe Market Technological Innovations and M&A Analysis
Technological innovations are playing a crucial role in the development of the Standard to Auto-Disable Syringe market. Manufacturers are continuously investing in research and development to improve the design and functionality of syringes. For example, the development of syringes with built-in safety features, such as auto-disable mechanisms and needle guards, has enhanced patient safety and reduced the risk of needlestick injuries. Additionally, advancements in materials science have led to the use of more durable and biocompatible materials in the production of syringes.
In terms of corporate activities, there has been a trend of mergers and acquisitions in the industry. Large companies are acquiring smaller firms to expand their product portfolios and gain a larger market share. For instance, Becton Dickinson’s acquisition of Bard in 2017 was a strategic move to strengthen its position in the medical device market. Such mergers and acquisitions not only enhance the capabilities of the acquiring company but also lead to economies of scale and improved efficiency in operations.
4 Global Standard to Auto-Disable Syringe Market Analysis by Type
The global market for Standard to Auto-Disable Syringes is anticipated to witness significant growth in the coming years, with distinct segments for Standard Syringes and Auto-Disable Syringes. Each type plays a crucial role in the healthcare sector, catering to different needs and preferences.
Standard Syringes are the conventional syringes widely used across various medical applications. They are essential tools in healthcare settings for tasks such as administering injections, drawing blood, and delivering medications. According to the forecast, the market size for Standard Syringes in 2025 is expected to reach 8,360.82 million USD. This represents a substantial portion of the total market, highlighting the ongoing demand for traditional syringes despite the advancements in medical technology.
The market for Standard Syringes is driven by their versatility and the familiarity of healthcare professionals with these devices. The forecasted growth from 2021 to 2025 indicates a steady increase, reflecting the consistent need for these syringes in routine medical practices.
Auto-Disable Syringes are designed with a safety feature that prevents the syringe from being used more than once, thus reducing the risk of infection transmission. These syringes are particularly valuable in public health initiatives and vaccination drives where the risk of cross-contamination is a significant concern. The market size for Auto-Disable Syringes in 2025 is projected to be 2,701.90 million USD.
Although this is a smaller segment compared to Standard Syringes, the growth rate is notable, indicating a rising awareness and adoption of safety measures in healthcare. The market for Auto-Disable Syringes is expected to grow as more healthcare facilities and professionals prioritize patient safety and adhere to guidelines that promote the use of auto-disable syringes.
Table Global Standard to Auto-Disable Syringe Market Size and Share by Type in 2025
|
Type |
Market Size (M USD) |
Market Share |
|---|---|---|
|
Standard Syringe |
8,360.82 |
73.03% |
|
Auto-Disable Syringe |
2,701.90 |
24.34% |
5 Global Standard to Auto-Disable Syringe Market Analysis by Application
The global market for Standard to Auto-Disable Syringes is anticipated to experience significant growth across various applications by 2025. The market is segmented into different applications, including Public Hospitals, Private Clinics, Nursing Homes, and Others.
Public hospitals are expected to be the largest consumer segment for Standard to Auto-Disable Syringes. In 2025, the market size for public hospitals is forecasted to reach 8,691.65 million USD. This represents a substantial increase from the 2021 figure of 7,018.30 million USD, indicating a strong growth trend. The growth in this segment can be attributed to the increasing demand for safe and effective medical procedures in public healthcare settings.
Private clinics are also a significant market segment, with a forecasted market size of 1,632.81 million USD in 2025. This reflects a growing trend towards private healthcare services and the need for reliable and safe medical devices in these settings. The market for private clinics has been steadily increasing, highlighting the importance of quality healthcare services in private practices.
Nursing homes are expected to see a market size of 360.35 million USD in 2025. This segment is particularly important due to the vulnerable population it serves. The elderly and those requiring long-term care often need regular medical attention, making the use of safe syringes crucial in these facilities. The forecasted growth in this segment underscores the increasing focus on elderly care and the need for safe medical practices.
Table Global Standard to Auto-Disable Syringe Market Size and Share by Application in 2025
|
Application |
Market Size (M USD) |
Market Share |
|---|---|---|
|
Public Hospital |
8,691.65 |
78.56% |
|
Private Clinic |
1,632.81 |
14.77% |
|
Nursing Home |
360.35 |
3.27% |
6 Global Standard to Auto-Disable Syringe Market Analysis by Region
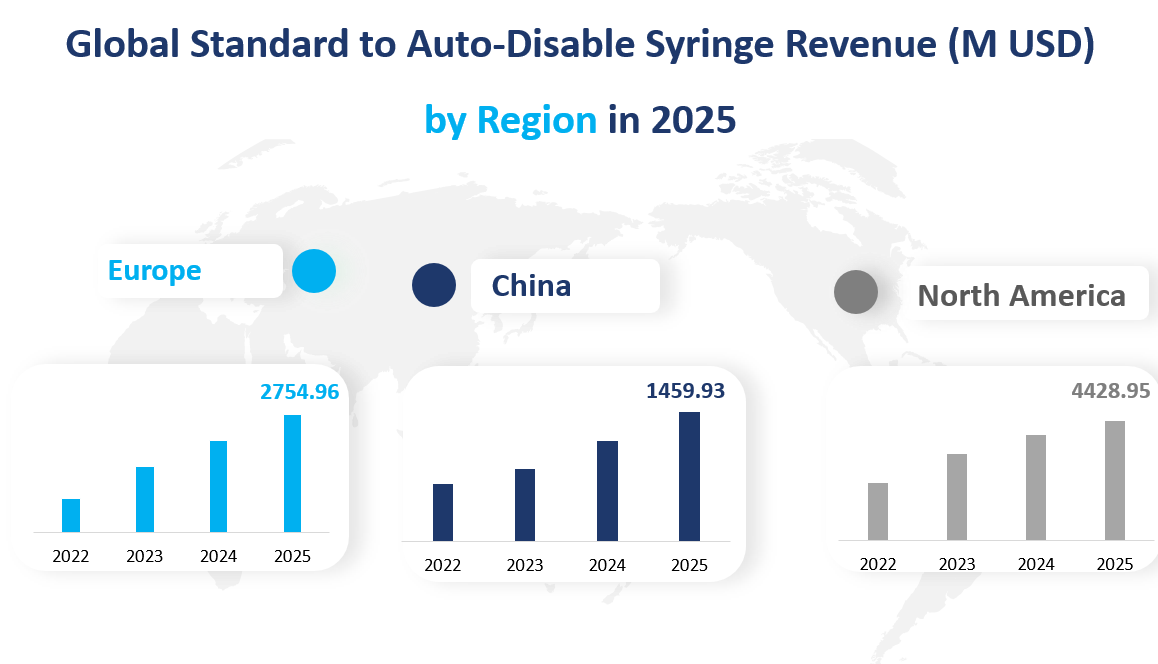
In 2025, North America is projected to be the largest regional market with a value of approximately 4,428.95 million USD. This region has consistently shown a strong demand for advanced medical devices, including Standard to Auto-Disable Syringes. The high growth rate can be attributed to factors such as the presence of major medical device manufacturers, a well-established healthcare infrastructure, and a focus on patient safety.
Europe is anticipated to be another significant market, with a value of around 2,754.96 million USD in 2025. The region has shown a growth rate of 6.55%, which is slightly lower than North America but still robust. The European market benefits from a combination of factors including a strong healthcare system, increasing awareness about the benefits of Auto-Disable Syringes, and supportive regulatory frameworks that facilitate the adoption of new medical technologies.
China is expected to be the fastest-growing region, with a market value of approximately 1,459.93 million USD in 2025. The growth rate in China is projected to be 7.96%, which is the highest among the analyzed regions. This rapid growth is driven by the country’s large population, increasing healthcare expenditure, and government initiatives to improve healthcare quality and access. Additionally, the rising middle class in China is more inclined towards investing in advanced healthcare solutions, further boosting the demand for Standard to Auto-Disable Syringes.
Figure Global Standard to Auto-Disable Syringe Revenue (M USD) by Region in 2025

7 Analysis of the Top 3 Companies in the Global Standard to Auto-Disable Syringe Market
7.1 Becton Dickinson
Company Introduction and Business Overview
Becton Dickinson, established in 1897, is a leading global medical technology company headquartered in the USA. Known for its extensive range of medical supplies, devices, and diagnostic products, Becton Dickinson serves healthcare institutions, life science researchers, and clinical laboratories worldwide. The company is committed to advancing the therapy for patients and improving operational efficiency for healthcare providers.
Products Offered
Becton Dickinson offers a comprehensive portfolio of Standard Syringes under its BD Integra™ brand. These syringes are designed for general use in medical settings, ensuring safety and reliability. The company’s syringes are available in various sizes and configurations, catering to diverse medical needs. Becton Dickinson’s focus on quality and innovation has made its syringes a preferred choice in hospitals and clinics globally.
7.2 Medtronic
Company Introduction and Business Overview
Founded in 1949, Medtronic is an American multinational medical technology company headquartered in Minnesota. The company is renowned for its medical devices, services, and solutions aimed at treating various chronic diseases and conditions. Medtronic serves patients and healthcare professionals worldwide, focusing on improving patient outcomes and enhancing clinical efficiency.
Products Offered
Medtronic provides Auto-Disable Syringes designed to prevent the reuse of syringes, thereby reducing the risk of infection transmission. Their syringes are used in a wide range of medical procedures, including vaccinations and medication administration. The company’s commitment to innovation is evident in its Auto-Disable Syringe technology, which ensures that once the syringe is used, it cannot be re-capped, thus enhancing patient safety.
7.3 Gerresheimer AG
Company Introduction and Business Overview
Established in 1864, Gerresheimer AG is a German multinational company specializing in pharmaceutical primary packaging. With a broad product range for pharmaceuticals, health, and well-being, Gerresheimer AG is a global partner for drug packaging and drug delivery systems. The company operates in Asia, Europe, and North America, serving a wide array of customers in the pharmaceutical, biotech, and healthcare sectors.
Products Offered
Gerresheimer AG offers Auto-Disable Syringes under its Gx RTF® ClearJect® Needle Syringes line. These syringes are designed to ensure safe and effective drug delivery, with sizes of 1.0 ml long and 2.25 ml available. The syringes are manufactured in a clean room environment according to GMP class C standards and are gamma sterilized, ensuring high-quality, reliable products. Gerresheimer AG’s focus on precision and safety in its syringes makes them a trusted choice in the healthcare industry.
1 Standard to Auto-Disable Syringe Introduction and Market Overview
1.1 Objectives of the Study
1.2 Overview of Standard to Auto-Disable Syringe
1.3 Standard to Auto-Disable Syringe Market Scope and Market Size Estimation
1.3.1 Market Concentration Ratio and Market Maturity Analysis
1.3.2 Global Standard to Auto-Disable Syringe Value and Growth Rate from 2020-2030
1.4 Market Segmentation
1.4.1 Types of Standard to Auto-Disable Syringe
1.4.2 Applications of Standard to Auto-Disable Syringe
1.4.3 Research Regions
1.5 Market Dynamics
1.5.1 Drivers
1.5.2 Limitations
1.5.3 Opportunities
1.6 Industry News and Policies by Regions
1.6.1 Industry News
1.6.2 Industry Policies
1.7 Standard to Auto-Disable Syringe Industry Development Trends under COVID-19 Outbreak
1.7.1 Global COVID-19 Status Overview
1.7.2 Influence of COVID-19 Outbreak on Standard to Auto-Disable Syringe Industry Development
2 Industry Chain Analysis
2.1 Upstream Raw Material Suppliers of Standard to Auto-Disable Syringe Analysis
2.2 Major Players Manufacturing Base of Standard to Auto-Disable Syringe in 2020
2.3 Standard to Auto-Disable Syringe Manufacturing Cost Structure Analysis
2.3.1 Production Process Analysis
2.3.2 Manufacturing Cost Structure of Standard to Auto-Disable Syringe
2.3.3 Raw Material Cost of Standard to Auto-Disable Syringe
2.3.4 Labor Cost of Standard to Auto-Disable Syringe
2.4 Market Channel Analysis of Standard to Auto-Disable Syringe
2.5 Major Downstream Buyers of Standard to Auto-Disable Syringe Analysis
3 Global Standard to Auto-Disable Syringe Market, by Type
3.1 Global Standard to Auto-Disable Syringe Value and Market Share by Type (2020-2025)
3.2 Global Standard to Auto-Disable Syringe Production and Market Share by Type (2020-2025)
3.3 Global Standard to Auto-Disable Syringe Value and Growth Rate by Type (2020-2025)
3.3.1 Global Standard to Auto-Disable Syringe Value and Growth Rate of Standard Syringe
3.3.2 Global Standard to Auto-Disable Syringe Value and Growth Rate of Auto-Disable Syringe
3.4 Global Standard to Auto-Disable Syringe Price Analysis by Type (2020-2025)
4 Standard to Auto-Disable Syringe Market, by Application
4.1 Downstream Market Overview
4.2 Global Standard to Auto-Disable Syringe Consumption and Market Share by Application (2020-2025)
4.3 Global Standard to Auto-Disable Syringe Consumption and Growth Rate by Application (2020-2025)
4.3.1 Global Standard to Auto-Disable Syringe Consumption and Growth Rate of Public Hospital (2020-2025)
4.3.2 Global Standard to Auto-Disable Syringe Consumption and Growth Rate of Private Clinic (2020-2025)
4.3.3 Global Standard to Auto-Disable Syringe Consumption and Growth Rate of Nursing Home (2020-2025)
5 Global Standard to Auto-Disable Syringe Production, Value (Million USD) by Region (2020-2025)
5.1 Global Standard to Auto-Disable Syringe Value and Market Share by Region (2020-2025)
5.2 Global Standard to Auto-Disable Syringe Production and Market Share by Region (2020-2025)
5.3 Global Standard to Auto-Disable Syringe Production, Value, Price and Gross Margin (2020-2025)
5.4 North America Standard to Auto-Disable Syringe Production, Value, Price and Gross Margin (2020-2025)
5.5 Europe Standard to Auto-Disable Syringe Production, Value, Price and Gross Margin (2020-2025)
5.6 China Standard to Auto-Disable Syringe Production, Value, Price and Gross Margin (2020-2025)
5.7 Japan Standard to Auto-Disable Syringe Production, Value, Price and Gross Margin (2020-2025)
5.8 Middle East and Africa Standard to Auto-Disable Syringe Production, Value, Price and Gross Margin (2020-2025)
5.9 India Standard to Auto-Disable Syringe Production, Value, Price and Gross Margin (2020-2025)
5.10 South America Standard to Auto-Disable Syringe Production, Value, Price and Gross Margin (2020-2025)
6 Global Standard to Auto-Disable Syringe Production, Consumption, Export, Import by Regions (2020-2025)
6.1 Global Standard to Auto-Disable Syringe Consumption by Regions (2020-2025)
6.2 North America Standard to Auto-Disable Syringe Production, Consumption, Export, Import (2020-2025)
6.3 Europe Standard to Auto-Disable Syringe Production, Consumption, Export, Import (2020-2025)
6.4 China Standard to Auto-Disable Syringe Production, Consumption, Export, Import (2020-2025)
6.5 Japan Standard to Auto-Disable Syringe Production, Consumption, Export, Import (2020-2025)
6.6 Middle East and Africa Standard to Auto-Disable Syringe Production, Consumption, Export, Import (2020-2025)
6.7 India Standard to Auto-Disable Syringe Production, Consumption, Export, Import (2020-2025)
6.8 South America Standard to Auto-Disable Syringe Production, Consumption, Export, Import (2020-2025)
7 Global Standard to Auto-Disable Syringe Market Status and SWOT Analysis by Regions
7.1 North America Standard to Auto-Disable Syringe Market Status and SWOT Analysis
7.1.1 North America Standard to Auto-Disable Syringe Market Under COVID-19
7.2 Europe Standard to Auto-Disable Syringe Market Status and SWOT Analysis
7.2.1 Europe Standard to Auto-Disable Syringe Market Under COVID-19
7.3 China Standard to Auto-Disable Syringe Market Status and SWOT Analysis
7.3.1 China Standard to Auto-Disable Syringe Market Under COVID-19
7.4 Japan Standard to Auto-Disable Syringe Market Status and SWOT Analysis
7.4.1 Japan Standard to Auto-Disable Syringe Market Under COVID-19
7.5 Middle East and Africa Standard to Auto-Disable Syringe Market Status and SWOT Analysis
7.5.1 Middle East and Africa Standard to Auto-Disable Syringe Market Under COVID-19
7.6 India Standard to Auto-Disable Syringe Market Status and SWOT Analysis
7.6.1 India Standard to Auto-Disable Syringe Market Under COVID-19
7.7 South America Standard to Auto-Disable Syringe Market Status and SWOT Analysis
7.7.1 South America Standard to Auto-Disable Syringe Market Under COVID-19
8 Competitive Landscape
8.1 Competitive Profile
8.2 Becton Dickinson
8.2.1 Company Profiles
8.2.2 Standard to Auto-Disable Syringe Product Introduction
8.2.3 Becton Dickinson Production, Value, Price, Gross Margin 2020-2025
8.3 Medtronic
8.3.1 Company Profiles
8.3.2 Standard to Auto-Disable Syringe Product Introduction
8.3.3 Medtronic Production, Value, Price, Gross Margin 2020-2025
8.4 Gerresheimer AG
8.4.1 Company Profiles
8.4.2 Standard to Auto-Disable Syringe Product Introduction
8.4.3 Gerresheimer AG Production, Value, Price, Gross Margin 2020-2025
8.5 Weigao Group
8.5.1 Company Profiles
8.5.2 Standard to Auto-Disable Syringe Product Introduction
8.5.3 Weigao Group Production, Value, Price, Gross Margin 2020-2025
8.6 Terumo Corporation
8.6.1 Company Profiles
8.6.2 Standard to Auto-Disable Syringe Product Introduction
8.6.3 Terumo Corporation Production, Value, Price, Gross Margin 2020-2025
8.7 Nipro Corporation
8.7.1 Company Profiles
8.7.2 Standard to Auto-Disable Syringe Product Introduction
8.7.3 Nipro Corporation Production, Value, Price, Gross Margin 2020-2025
8.8 B. Braun Medical
8.8.1 Company Profiles
8.8.2 Standard to Auto-Disable Syringe Product Introduction
8.8.3 B. Braun Medical Production, Value, Price, Gross Margin 2020-2025
8.9 Hindustan Syringes & Medical Devices Ltd
8.9.1 Company Profiles
8.9.2 Standard to Auto-Disable Syringe Product Introduction
8.9.3 Hindustan Syringes & Medical Devices Ltd Production, Value, Price, Gross Margin 2020-2025
8.10 Baxter International
8.10.1 Company Profiles
8.10.2 Standard to Auto-Disable Syringe Product Introduction
8.10.3 Baxter International Production, Value, Price, Gross Margin 2020-2025
8.11 AlShifa Medical
8.11.1 Company Profiles
8.11.2 Standard to Auto-Disable Syringe Product Introduction
8.11.3 AlShifa Medical Products Production, Value, Price, Gross Margin 2020-2025
8.12 Smiths Medical
8.12.1 Company Profiles
8.12.2 Standard to Auto-Disable Syringe Product Introduction
8.12.3 Smiths Medical Production, Value, Price, Gross Margin 2020-2025
8.13 Kindly Group
8.13.1 Company Profiles
8.13.2 Standard to Auto-Disable Syringe Product Introduction
8.13.3 Kindly Group Production, Value, Price, Gross Margin 2020-2025
8.14 Fresenius Kabi AG
8.14.1 Company Profiles
8.14.2 Standard to Auto-Disable Syringe Product Introduction
8.14.3 Fresenius Kabi AG Production, Value, Price, Gross Margin 2020-2025
8.15 VOGT Medical
8.15.1 Company Profiles
8.15.2 Standard to Auto-Disable Syringe Product Introduction
8.15.3 VOGT Medical Production, Value, Price, Gross Margin 2020-2025
8.16 Iscon Surgicals
8.16.1 Company Profiles
8.16.2 Standard to Auto-Disable Syringe Product Introduction
8.16.3 Iscon Surgicals Production, Value, Price, Gross Margin 2020-2025
9 Global Standard to Auto-Disable Syringe Market Analysis and Forecast by Type and Application
9.1 Global Standard to Auto-Disable Syringe Market Value & Volume Forecast, by Type (2025-2030)
9.1.1 Standard Syringe Market Value and Volume Forecast (2025-2030)
9.1.2 Auto-Disable Syringe Market Value and Volume Forecast (2025-2030)
9.2 Global Standard to Auto-Disable Syringe Market Value & Volume Forecast, by Application (2025-2030)
9.2.1 Public Hospital Market Value and Volume Forecast (2025-2030)
9.2.2 Private Clinic Market Value and Volume Forecast (2025-2030)
9.2.3 Nursing Home Market Value and Volume Forecast (2025-2030)
10 Standard to Auto-Disable Syringe Market Analysis and Forecast by Region
10.1 North America Market Value and Consumption Forecast (2025-2030)
10.2 Europe Market Value and Consumption Forecast (2025-2030)
10.3 China Market Value and Consumption Forecast (2025-2030)
10.4 Japan Market Value and Consumption Forecast (2025-2030)
10.5 Middle East and Africa Market Value and Consumption Forecast (2025-2030)
10.6 India Market Value and Consumption Forecast (2025-2030)
10.7 South America Market Value and Consumption Forecast (2025-2030)
10.8 Standard to Auto-Disable Syringe Market Forecast Under COVID-19
11 New Project Feasibility Analysis
11.1 Industry Barriers and New Entrants SWOT Analysis
12 Research Finding and Conclusion
13 Appendix
13.1 Methodology
13.2 Research Data Source
